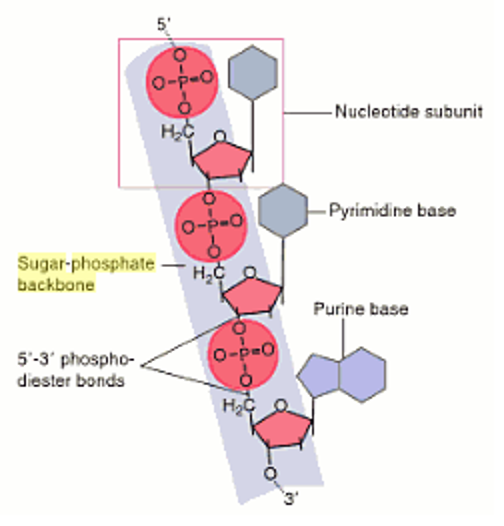
Artificial analogues with 2′-ribose modifications such as 2′-fluoro, 2′- O-methyl, 2′- O-methoxyethyl, and locked nucleic acids (LNAs) are used in the nucleic acid-based drugs and drug candidates 9, 10, 11, 12, 13, 14. In the past decades, many nucleic acid analogues have been developed. They have potential for use in nucleic acid-based drugs, in development of artificial genetic polymers, and in the prebiotic field 1, 2, 3, 4, 5, 6, 7, 8. Xeno nucleic acids (XNAs) are synthetic analogues that retain natural nucleobases but are replaced with backbone structures different from DNA and RNA.  
The unique structural features of the RNA-recognizing mode of L- aTNA and SNA should prove useful in nanotechnology, biotechnology, and basic research into prebiotic chemistry. In addition, SNA and L- aTNA form a triplex structure via C:G*G parallel Hoogsteen interactions with RNA. The unwound helical structures originate from interactions between nucleobases and neighbouring backbones of L- aTNA and SNA through CH–O bonds. Unlike canonical A-type duplexes, the base pairs in the heteroduplexes align perpendicularly to the helical axes, and consequently helical pitches are large. The heteroduplexes show unwound right-handed helical structures. Here we disclose crystal structures of RNA hybridizing with SNA and with L- aTNA. Although few acyclic nucleic acids can stably bind to RNA and DNA, serinol nucleic acid (SNA) and L-threoninol nucleic acid (L- aTNA) stably bind to them. Xeno nucleic acids, which are synthetic analogues of natural nucleic acids, have potential for use in nucleic acid drugs and as orthogonal genetic biopolymers and prebiotic precursors.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
